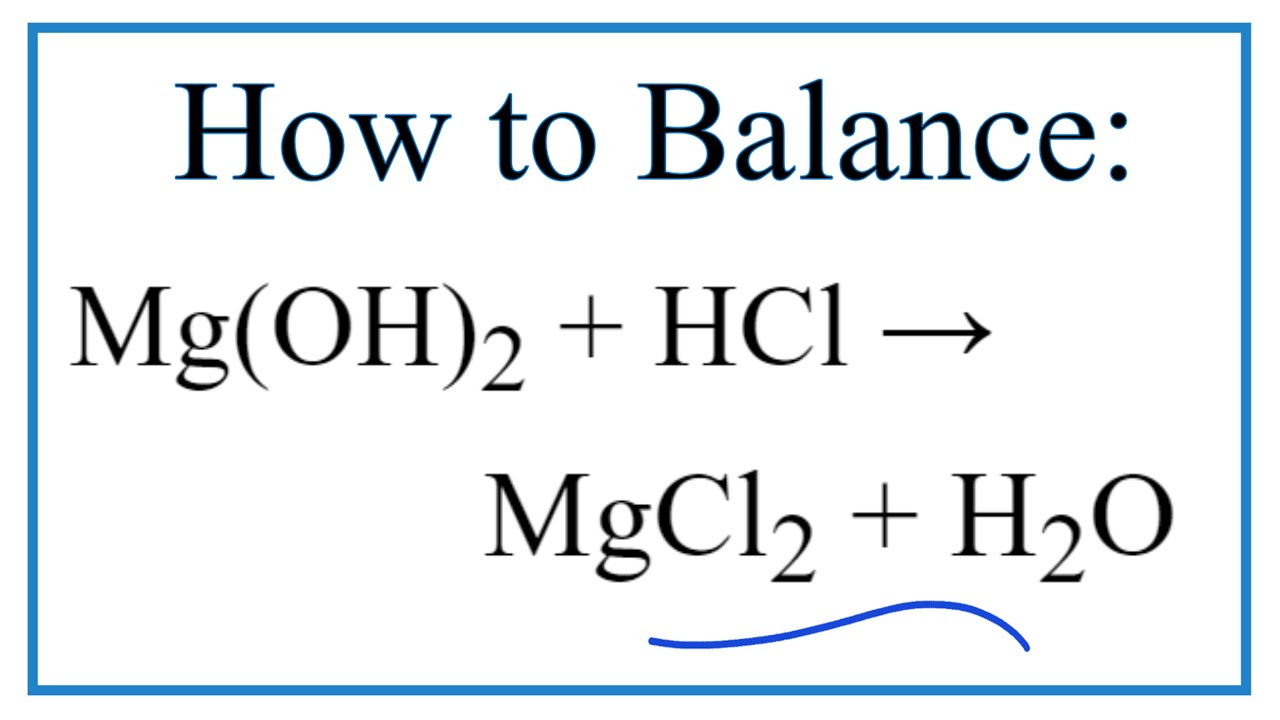
SOLVED: When hydrochloric acid reacts with magnesium hydroxide, the products that form are magnesium chloride and water. What is the coefficient for water when this equation is properly balanced?
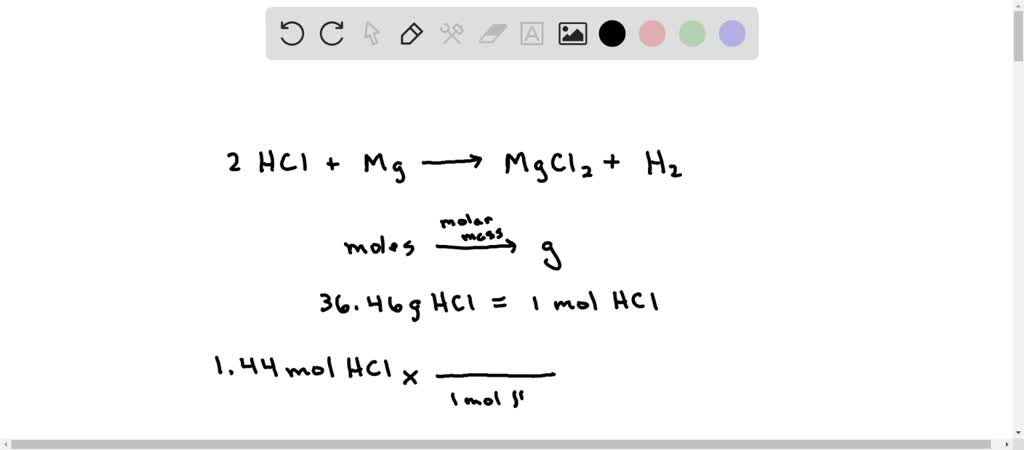
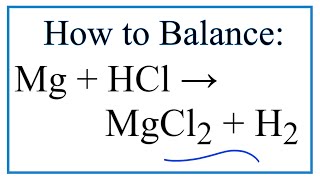
SOLVED: When magnesium metal (Mg) reacts with hydrochloric acid (HCl), magnesium chloride (MgCl2) and hydrogen gas (H2) are produced according to the following chemical equation. 2 HCl (aq) + Mg (s) →

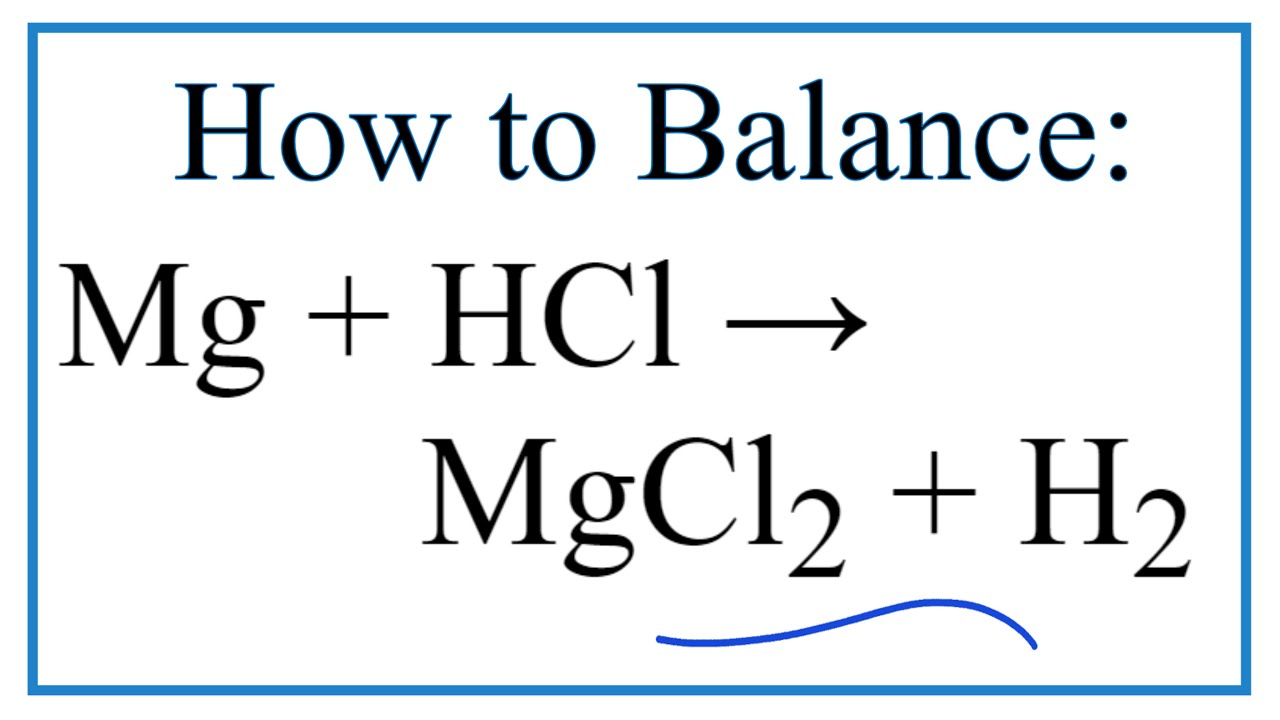
How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) | How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) One of the

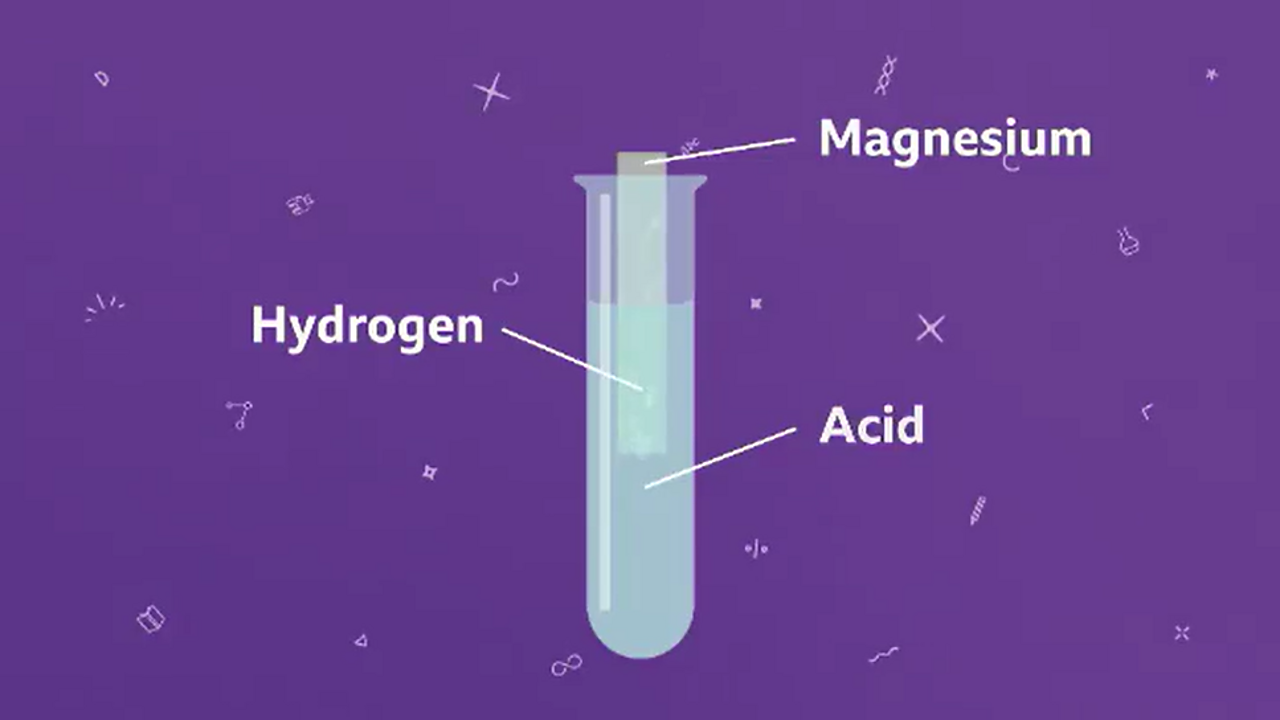
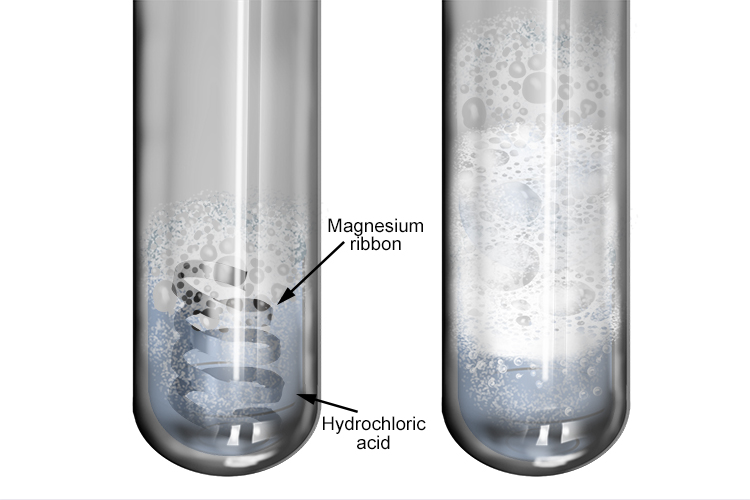
Describe an experiment to study the speed of reaction between dilute hydrochloric acid and magnesium, by measuring the volume of gas produced over time. - Study notes, tips, worksheets, exam papers