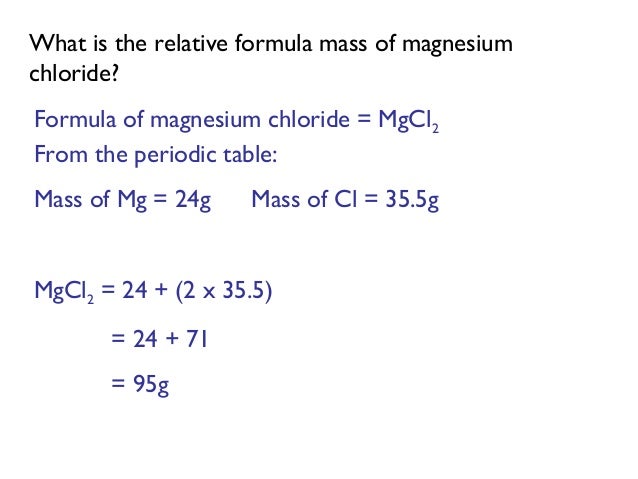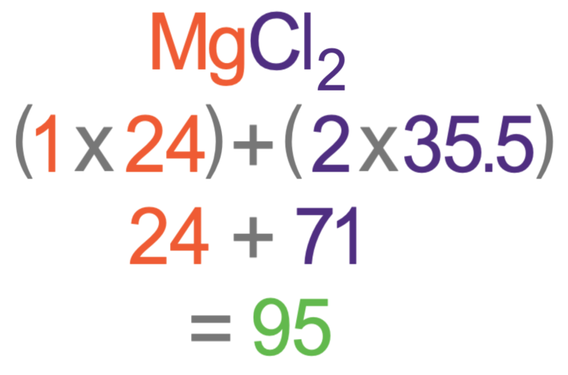Calculate the Molar Mass Magnesium chloride Ammonium sulfate Answers: g/mol; g/mol. - ppt download

How many moles of magnesium chloride are formed when 55 mL of 0.70 M HCl is added to 42 mL of 1.204 g Mg(OH)_2? Mg(OH)_2(s) + 2HCl(aq) \rightarrow MgCl_2(aq) + 2H_2O(l) A.

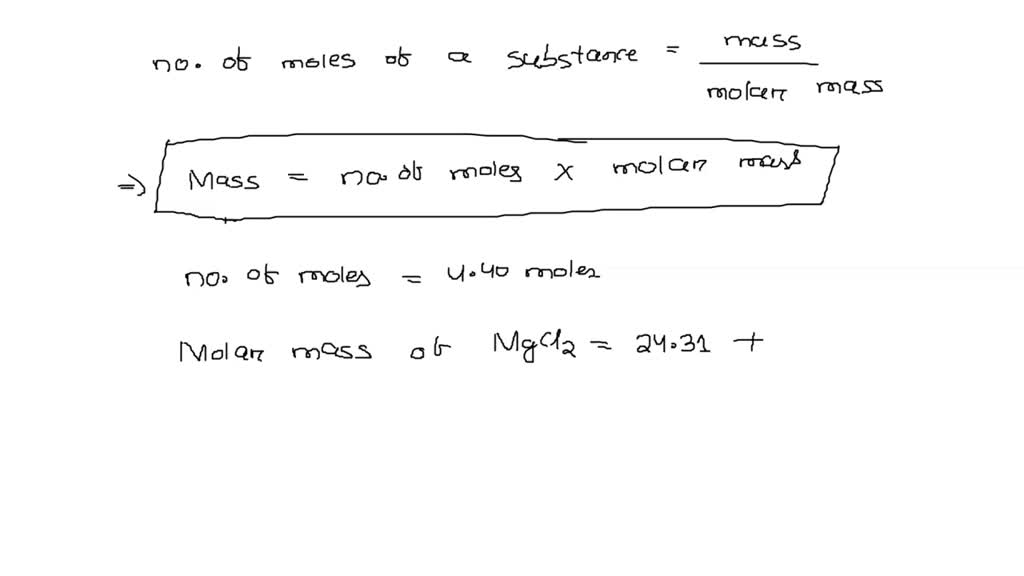
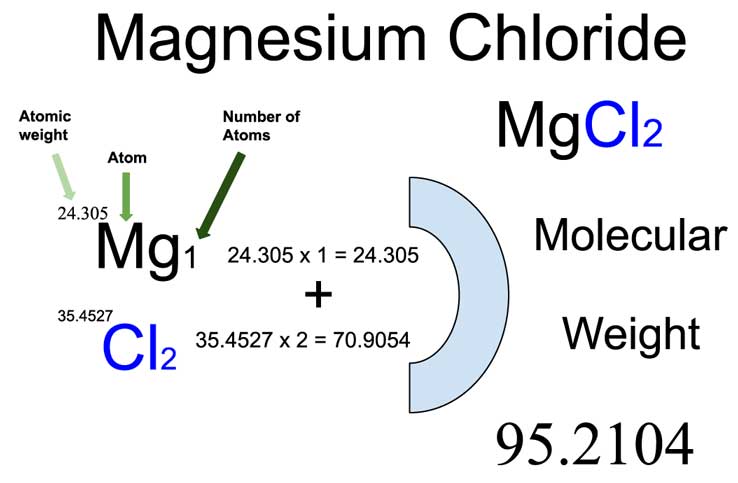
Write the names of the following compounds and deduce their molar masses. Na2SO4, K2CO3, CO2, MgCl2, NaOH, AlPO4, NaHCO3

Write the formula for the following and calculate the molecular mass for each one of them.1). Potassium - Brainly.in